Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hirooka, Shun; Yokoyama, Keisuke; Kato, Masato
Proceedings of International Conference on Fast Reactors and Related Fuel Cycles; Sustainable Clean Energy for the Future (FR22) (Internet), 8 Pages, 2022/04
Property studies on Am/Np-bearing MOX were carried out and how the properties influences on the irradiation behaviors was discussed. Both Am and Np inclusions increase the oxygen potential of MOX. Inter-diffusion coefficients obtained by using diffusion couple technique indicate that the inter-diffusion coefficient is larger in the order of U-Am, U-Pu and U-Np. Also, the inter-diffusion coefficients were evaluated to be larger at the O/M = 2 than those of O/M  2 by several orders. The increase of oxygen potential with Am/Np leads to higher vapor pressure of UO
2 by several orders. The increase of oxygen potential with Am/Np leads to higher vapor pressure of UO and the acceleration of the pore migration along temperature gradient during irradiation. The redistributions of actinide elements were also considered with the relationship of the pore migration and diffusion in solid state. Thus, the obtained inter-diffusion coefficients directly influence on the redistribution rate. The obtained properties were modelled and can be installed in a fuel irradiation simulation code.
and the acceleration of the pore migration along temperature gradient during irradiation. The redistributions of actinide elements were also considered with the relationship of the pore migration and diffusion in solid state. Thus, the obtained inter-diffusion coefficients directly influence on the redistribution rate. The obtained properties were modelled and can be installed in a fuel irradiation simulation code.
Yamaguchi, Tetsuji; Ohira, Saki; Hemmi, Ko; Barr, L.; Shimada, Asako; Maeda, Toshikatsu; Iida, Yoshihisa
Radiochimica Acta, 108(11), p.873 - 877, 2020/11
Times Cited Count:7 Percentile:66.68(Chemistry, Inorganic & Nuclear)Tachi, Yukio; Suyama, Tadahiro*; Mihara, Morihiro
JAEA-Data/Code 2019-021, 101 Pages, 2020/03
Sorption of radionuclides in cement and bentonite as engineered barrier materials, and rocks as natural barrier is the one of key processes in the performance assessment of geological disposal of TRU and high-level waste. The magnitude of sorption, expressed normally by a distribution coefficient (K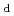 ), needs to be measured and determined taking into account the properties of barrier materials and geochemical conditions and associated uncertainty in the performance assessment. The basic concept for TRU waste disposal contains cementitious materials as an engineered barrier materials, in addition to bentonite and rock. It is therefore needed to consider the effects of the cement degradation and co-existing substances such as nitrates on radionuclide sorption. This report focused on data acquisition of distribution coefficient (K
), needs to be measured and determined taking into account the properties of barrier materials and geochemical conditions and associated uncertainty in the performance assessment. The basic concept for TRU waste disposal contains cementitious materials as an engineered barrier materials, in addition to bentonite and rock. It is therefore needed to consider the effects of the cement degradation and co-existing substances such as nitrates on radionuclide sorption. This report focused on data acquisition of distribution coefficient (K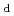 ) by batch sorption experiments for the systems coupling barrier material-chemical condition-radionuclides that are needed to consider for the performance assessment of geological disposal of TRU waste. The barrier materials considered are ordinary Portland cement (OPC), degraded OPC and tuff rock. The chemical conditions are distilled water and synthetic seawater equilibrated with OPC and those containing nitrates and ammonium salts, etc. The radionuclides considered are organic carbon, inorganic carbon, Cl, I, Cs, Ni, Se, Sr, Sn, Nb, Am and Th. Although K
) by batch sorption experiments for the systems coupling barrier material-chemical condition-radionuclides that are needed to consider for the performance assessment of geological disposal of TRU waste. The barrier materials considered are ordinary Portland cement (OPC), degraded OPC and tuff rock. The chemical conditions are distilled water and synthetic seawater equilibrated with OPC and those containing nitrates and ammonium salts, etc. The radionuclides considered are organic carbon, inorganic carbon, Cl, I, Cs, Ni, Se, Sr, Sn, Nb, Am and Th. Although K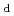 values have been partly reported previously as RAMDA (Radionuclide Migration Datasets) for the performance assessment in the TRU-2 report, these results and addition K
values have been partly reported previously as RAMDA (Radionuclide Migration Datasets) for the performance assessment in the TRU-2 report, these results and addition K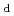 data are reported with the details of experimental methods and conditions.
data are reported with the details of experimental methods and conditions.
Iida, Yoshihisa; Yamaguchi, Tetsuji; Tanaka, Tadao; Hemmi, Ko
Journal of Nuclear Science and Technology, 53(10), p.1573 - 1584, 2016/10
Times Cited Count:10 Percentile:64.88(Nuclear Science & Technology)The sorption behavior of thorium (Th) onto granitic rock and its major constituent were investigated by batch sorption experiments. Experiments were carried out under variable pH and carbonate concentrations. Distribution coefficients decreased with increased carbonate concentrations and showed the minimal value at pH 9-10. This sorption tendency was likely due to forming the hydroxide-carbonate complexes of Th in the solutions. The order of sorbability for Th was mica  feldspar
feldspar  quartz = granite. The sorption behaviors of Th onto these minerals were analyzed by the triple-layer surface complexation model with the Visual Minteq computer program. The model calculations assuming the inner-sphere surface complexation of Th were able to explain the experimental results reasonably well. It was shown that the sorption behavior of Th onto granite can be explained primarily by the complexation with the surface sites of feldspar.
quartz = granite. The sorption behaviors of Th onto these minerals were analyzed by the triple-layer surface complexation model with the Visual Minteq computer program. The model calculations assuming the inner-sphere surface complexation of Th were able to explain the experimental results reasonably well. It was shown that the sorption behavior of Th onto granite can be explained primarily by the complexation with the surface sites of feldspar.
Yamaguchi, Tetsuji; Nakayama, Shinichi; Yoshida, Takahiro
Radiochimica Acta, 92(9-11), p.677 - 682, 2004/12
Times Cited Count:6 Percentile:40.72(Chemistry, Inorganic & Nuclear)Adsorption of actinides onto negatively charged mineral surfaces was investigated under conditions that actinides were predominantly present as anionic complex species: Th(CO )
)
 , Am(CO
, Am(CO )
)
 , Np(CO
, Np(CO )
) (OH)
(OH)
 , UO
, UO (OH)
(OH)
 , NpO
, NpO (OH)
(OH)
 , Sn(OH)
, Sn(OH)
 and Pb(OH)
and Pb(OH)
 . These solutions were left to stand for 2 days to confirm these elements stable in dissolved state, and then contacted with minerals,
. These solutions were left to stand for 2 days to confirm these elements stable in dissolved state, and then contacted with minerals,  -Al
-Al O
O or SiO
or SiO (AEROSIL, specific surface area: 10
(AEROSIL, specific surface area: 10 m
m kg
kg ). After desired contact time for 2 days or more, the solutions were ultra-filtered through 10
). After desired contact time for 2 days or more, the solutions were ultra-filtered through 10 -molecular-weight cutoff Millipore filters and the concentrations of the elements in the filtrates were determined. The sorption experiments were performed at room temperature (25
-molecular-weight cutoff Millipore filters and the concentrations of the elements in the filtrates were determined. The sorption experiments were performed at room temperature (25 C) under Ar. Distribution coefficients decreased with the increasing pH and with increasing carbonate concentrations. The monotonous decrease in the distribution coefficients in the investigated pH range suggests that the electrostatic repulsion was a dominant interaction between anionic complex species of actinides and negatively charged mineral surfaces.
C) under Ar. Distribution coefficients decreased with the increasing pH and with increasing carbonate concentrations. The monotonous decrease in the distribution coefficients in the investigated pH range suggests that the electrostatic repulsion was a dominant interaction between anionic complex species of actinides and negatively charged mineral surfaces.
Nojiri, Naoki; Shimakawa, Satoshi; Fujimoto, Nozomu; Goto, Minoru
Nuclear Engineering and Design, 233(1-3), p.283 - 290, 2004/10
Times Cited Count:12 Percentile:61.53(Nuclear Science & Technology)This paper describes the results of core physics test in start-up and power-up of the HTTR. The tests were conducted in order to ensure performance and safety of the high temperature gas cooled reactor, and was carried out to measure the critical approach, the excess reactivity, the shutdown margin, the control rod worth, the reactivity coefficient, the neutron flux distribution and the power distribution. The expected core performance and the required reactor safety characteristics were verified from the results of measurements and calculations.
 Np and
Np and  Am through loess media
Am through loess mediaTanaka, Tadao; Mukai, Masayuki; Maeda, Toshikatsu; Matsumoto, Junko; Ogawa, Hiromichi; Li, Z.*; Wang, X.*; Fan, Z.*; Guo, L.*; Liu, C.*
Journal of Radioanalytical and Nuclear Chemistry, 256(2), p.205 - 211, 2003/05
Times Cited Count:3 Percentile:25.79(Chemistry, Analytical)Migration experiments of  Np(V) and
Np(V) and  Am(III) have been performed using a column system, to investigate migration behavior of Np and Am through a column packed with loess, taken from Shanxi, China. Adsorption mechanisms of Np and Am on the loess were examined by a chemical extraction method. In the case of the Np, most of Np adsorbed on the influent edge of the column. The Np adsorbed on the loess was mainly controlled by surface complexation. However, the migration of Np in the loess media could be roughly evaluated by using the distribution coefficient. In the case of the Am, particulate Am species was formed in the influent solution and moved in the column. The Am adsorbed on the loess was controlled by irreversible reactions. The migration behavior of particulate Am in the loess media could be expressed by the filtration theory.
Am(III) have been performed using a column system, to investigate migration behavior of Np and Am through a column packed with loess, taken from Shanxi, China. Adsorption mechanisms of Np and Am on the loess were examined by a chemical extraction method. In the case of the Np, most of Np adsorbed on the influent edge of the column. The Np adsorbed on the loess was mainly controlled by surface complexation. However, the migration of Np in the loess media could be roughly evaluated by using the distribution coefficient. In the case of the Am, particulate Am species was formed in the influent solution and moved in the column. The Am adsorbed on the loess was controlled by irreversible reactions. The migration behavior of particulate Am in the loess media could be expressed by the filtration theory.
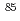 Sr,
Sr,  Np,
Np,  Pu and
Pu and  Am in loess media (Joint research)
Am in loess media (Joint research)Tanaka, Tadao; Mukai, Masayuki; Maeda, Toshikatsu; Matsumoto, Junko; Ogawa, Hiromichi; Li, S.*; Wang, Z.*; Wang, J.*; Guo, Z.*; Zhao, Y.*
JAERI-Research 2002-034, 20 Pages, 2002/12
Adsorption mechanisms and models of 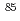 Sr(II),
Sr(II),  Np(V),
Np(V),  Pu(IV) and
Pu(IV) and  Am(III) on the loess were investigated from their adsorption and desorption properties. The distribution coefficient of
Am(III) on the loess were investigated from their adsorption and desorption properties. The distribution coefficient of 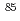 Sr and
Sr and  Np was 2 - 3 orders of magnitude smaller than that of
Np was 2 - 3 orders of magnitude smaller than that of  Pu and
Pu and  Am. The adsorption of
Am. The adsorption of 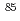 Sr and
Sr and  Np was mainly controlled by the ion exchange reaction. On the other hand, the adsorption of
Np was mainly controlled by the ion exchange reaction. On the other hand, the adsorption of  Pu and
Pu and  Am was mostly controlled by the selective chemical reactions with Fe and Mn oxyhydroxide/oxide and humic substances. On the basis of the experimental results, several types of adsorption models of the radionuclides, considering elemental concentrations, adsorption mechanisms and kinetics, were proposed for setting up the analytical systems of radionuclide migration in the loess media.
Am was mostly controlled by the selective chemical reactions with Fe and Mn oxyhydroxide/oxide and humic substances. On the basis of the experimental results, several types of adsorption models of the radionuclides, considering elemental concentrations, adsorption mechanisms and kinetics, were proposed for setting up the analytical systems of radionuclide migration in the loess media.
; Usuda, Shigekazu
Journal of Alloys and Compounds, 271-273, p.244 - 247, 1998/00
Times Cited Count:17 Percentile:69.26(Chemistry, Physical)no abstracts in English
Takahashi, Tomoyuki; Takebe, Shinichi; Kimura, Hideo; Matsuzuru, Hideo; Yasuda, Hiroshi*; Uchida, Shigeo*; Saeki, Akiyoshi*; Mahara, Yasunori*; Sasaki, Noriyuki*; *; et al.
JAERI-Research 97-089, 25 Pages, 1997/12
no abstracts in English
Kato, Shohei; Yanase, Yoshiaki; Honda, Tetsutaro*
IRPA9: 1996 International Congress on Radiation Protection, Proceedings, 3, p.354 - 356, 1996/00
no abstracts in English
Kato, Shohei; Yanase, Yoshiaki
JAERI-M 93-113, 17 Pages, 1993/05
no abstracts in English
Fujine, Sachio; Uchiyama, Gunzo; Maeda, Mitsuru; R.Thompson*; C.Mason*; R.Bush*
Solvent Extraction in the Process Industries, Vol. 3, p.1789 - 1796, 1993/00
no abstracts in English
; ; Hoshiya, Taiji
Journal of Nuclear Science and Technology, 28(10), p.961 - 964, 1991/10
no abstracts in English
; ; Takeda, Tsuneo; Muraoka, Susumu;
Hoshasei Haikibutsu Kenkyu Renrakukai Rombunshu, VI, p.64 - 75, 1991/00
no abstracts in English
JAERI-M 87-063, 133 Pages, 1987/04
no abstracts in English
; ; ; ; ;
Nucl.Eng.Des., 102, p.11 - 20, 1987/00
Times Cited Count:5 Percentile:50.76(Nuclear Science & Technology)no abstracts in English
;
Nihon Genshiryoku Gakkai-Shi, 28(4), p.344 - 351, 1986/00
Times Cited Count:0 Percentile:0.02(Nuclear Science & Technology)no abstracts in English
; ; *
JAERI-M 85-186, 58 Pages, 1985/11
no abstracts in English
; Wadachi, Yoshiki
JAERI-M 8044, 19 Pages, 1979/01
no abstracts in English